The European healthcare landscape is a dynamic, innovative ecosystem. Industry is increasingly identifying new approaches, solutions and business models that adapt to both mature and emerging growth markets. The pace of innovation is relentless, the potential benefits for society many.
At the same time, this is a highly regulated industry, working to the highest possible standards of safety and trust. Regulatory pathways are also evolving to adapt to the challenges posed by new therapies, new delivery methods and new health challenges.
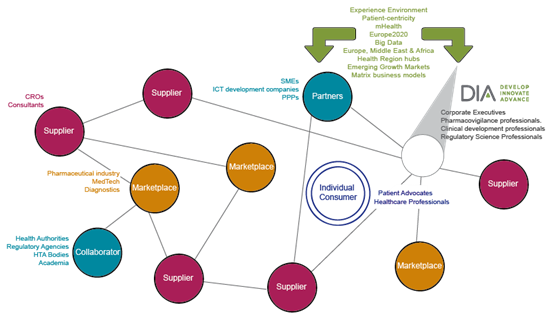
To maintain user-driven innovation as an attractive investment for all stakeholders, the value chain needs to be ever more cohesive. Healthcare innovators, decision makers, regulators and payers must to work together to shape their environment. Future barriers should be minimised, potential benefits to society maximised and treatments delivered as quickly as possible to the patients that need them.

EuroMeeting 2015
The DIA EuroMeeting 2015 offers the ideal platform for healthcare innovators, patient advocates, decision makers and regulatory professionals to connect. They can network, scope potential new business partnerships and explore options for concrete solutions. This is why the EuroMeeting 2015 offers a significant return on investment for companies seeking to proactively manage a successful shift to a service oriented model.
How will you benefit?
Your core staff will not only learn about current and future market conditions issues. They can also spend time actively networking and building relationships with the regulatory and policy counterparts who are shaping trends. The resulting knowledge transfer and lasting partnerships will reduce the burden of regulatory barriers, benefiting your business now and further down the line.
Who will you meet?
The EuroMeeting attracts:
• Corporate Executives
• Pharmacovigilance professionals
• Clinical development professionals
• Regulators
• Decision Makers
• CRO’s/Consultants
What makes the EuroMeeting 2015 different?
The DIA EuroMeeting 2015 is unique in presenting a microcosm of the entire drug development process ranging from the policy environment to market uptake and regulation. It attracts attendees & exhibitors from all stages of the value chain, ranging from those conducting original R&D to the patients benefiting from innovation
– See more at: http://www.diahome.org/en/Flagship-Meetings/EuroMeeting2015/About-the-Conference.aspx#sthash.TxfQPRV1.dpuf
In an intense and stimulating three days, you will not only learn, you will be exposed to fresh thinking and the latest ideas. You will meet not only people you know, but also those that you want to know and those that you should know. – See more at: http://www.diahome.org/en/Flagship-Meetings/EuroMeeting2015/About-the-Conference.aspx#sthash.TxfQPRV1.dpuf
13 patients were attending this conference through the fellowship grant program of DIA. Vesna Aleksovska as a representative of Life With Challenges attended the conference. Patients interested in attending the conference next year are welcome to contact Life With Challenges and ask for information about applying for the fellowship grant.